Diseases investigated at the Institut du Cerveau – ICM: Amyotrophic lateral sclerosis, Parkinson’s disease and Movement disorders, Huntington’s disease, Alzheimer’s and Prion diseases, Multiple sclerosis, Neuro-oncology, Epilepsy, Autism, Depression, Frontotemporal Lobar degenerations, Tourette syndrom, Sleep disorders.
Centre d’Investigation Clinique Neurosciences (CIC Neurosciences) consists in a full pipeline of clinical research facilities dedicated to neuroscience at Institut du Cerveau – ICM and within the hospital , from bench to bedside. It is located in two different areas within Pitié-Salpêtrière hospital : the Institut du Cerveau – ICM building, and Paul Castaigne neurological department.
ROLE
- organizing a unique entry for clinical trials for investigators at Institut du Cerveau – ICM and Pôle MSN;
- coordinating clinical research trials in neuroscience : proof-of-concept, phase II, phase III, cohort and translational studies;
- provide the amenities to ensure high quality and ethic in clinical trials, and safety of volunteers participating in trials.
 CLINICAL TRIALS
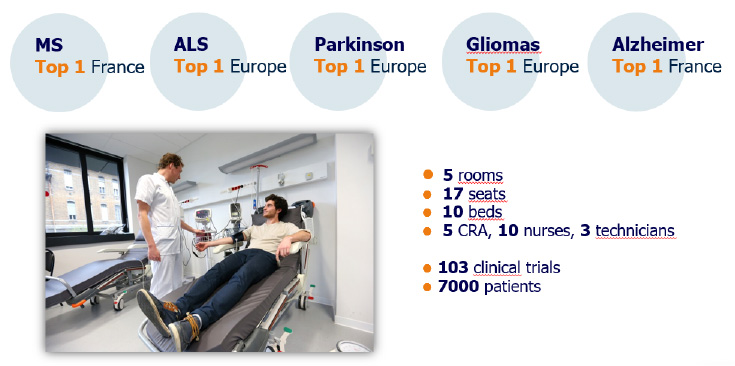
CLINICAL TRIALS
- more than 70 ongoing trials in neuroscience (in 2017),
- more than 2000 visits per year (in 2017),
- In addition, partnerships with industrial companies for early drug development were achieved.
Development of medical devices
The interaction between the CIC with the Living Lab and the start-ups/iPEPS incubated at the Institut du Cerveau – ICM were considerably increased, particularly for the development of medical devices. The AP-HP partner is essential to provide an extensive technical medical infrastructure by stabilizing clinical personnel dedicated to clinical research.
CHALLENGES
A working group on clinical and translational research has started reflecting on how to : 1) optimize the organization of clinical research in the field of neurosciences at the Pitié-Salpêtrière; 2) foster the translational research for a stronger impact on clinical practice; 3) develop early clinical trials with a professional and dedicated team; 4) increase large-scale cohorts of patients including clinical/imaging/physiology data coming from the clinical environment and address data sharing issues. This working group included clinicians from the Pôle des Maladies du Système Nerveux and researchers from the Institut du Cerveau – ICM and led to very fruitful discussions and proposals for the coming years at the Institut du Cerveau – ICM.
EXAMPLES
- Creation of a unique early Phase clinical trial Center (Institut du Cerveau – ICM@NEUROTRIALS)
- NeuroCatalyst: accelerate product development